 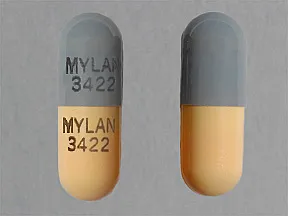 
The remaining 75% is nitrofurantoin monohydrate contained in a powder blend which, upon exposure to gastric and intestinal fluids, forms a gel matrix that releases nitrofurantoin over time. Twenty-five percent is macrocrystalline nitrofurantoin, which has slower dissolution and absorption than nitrofurantoin monohydrate. 10, FD&C Red No.40, gelatin, titanium dioxide, lactose, magnesium stearate, povidone, talc, sodium lauryl sulfate, colloidal silicon dioxide and Opacode® black ink S-1-17843 (consist of shellac, ferrosoferric oxide, butyl alcohol, propylene glycol, isopropyl alcohol and ammonia).įDA approved dissolution test specifications differ from USP.Įach Nitrofurantoin Capsules, USP (monohydrate/ macrocrystals) contains two forms of nitrofurantoin. Inactive Ingredients: Each capsule contains carbomer 974P, pregelatinized starch, sucrose, FD&C Blue No.1, FD&C Yellow No. The chemical name of nitrofurantoin monohydrate is 1-methylene]amino]-2,4-imidazolidinedione monohydrate. The chemical name of nitrofurantoin macrocrystals is 1-methylene]amino]-2,4- imidazolidinedione. , USP (monohydrate/macrocrystals)is a hard gelatin capsule shell containing the equivalent of 100 mg of nitrofurantoin in the form of 25 mg of nitrofurantoin macrocrystals and 75 mg of nitrofurantoin monohydrate. Nitrofurantoin is an antibacterial agent specific for urinary tract infections.
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
